Introduction
For the last project of our junior year, we were given a very broad and open opportunity to create our very own chemistry project. This would reflect the laws of thermodynamics and other related content we had recently learned. We were given this assignment after learning multiple pieces of information about power and energy. Contrary to our usual classroom dynamic, this unit was done differently than others, because of the remote learning due to the Covid-19 pandemic.
I, myself, chose to participate in my project on my own with the help of my brother as my experiment partner. I wanted to turn this project into something that could cure my boredom from lock down, while simultaneously displaying the Laws of Thermodynamics. It was through these guidelines that I decided to create a baking soda and vinegar bottle rocket out of common household items and hold a competition with my brother.
I, myself, chose to participate in my project on my own with the help of my brother as my experiment partner. I wanted to turn this project into something that could cure my boredom from lock down, while simultaneously displaying the Laws of Thermodynamics. It was through these guidelines that I decided to create a baking soda and vinegar bottle rocket out of common household items and hold a competition with my brother.
Vocabulary
1st Law of Thermodynamics- energy cannot be created or destroyed, only converted into other forms. This is the whole idea behind this project because the chemical energy isn't lost, it is changed into thermal energy.
Ionic Solution- a solution that is filled with ions. Ionic compounds separate into cations and anions when in the solution. The sodium acetate in water is the ionic solution in this project.
Phase Change- when a substance changes from one state to another. The three most common states are solid, liquid, and gas. One example of a phase change is ice (solid) melting into water (liquid).
Exothermic- when something is exothermic it releases or gives off energy. During the phase change, the hand warmer is releasing the energy it needed to separate the ionic compound making it exothermic.
Thermal Energy- A type of energy that is given as heat. All heat that exists is thermal energy. It is released when the phase change occurs in this project.
Energy Transfer- when energy changes from one type to another. All energy is conserved but can change between the different types. Some examples are mechanical, chemical, and electrical energy.
Ionic Solution- a solution that is filled with ions. Ionic compounds separate into cations and anions when in the solution. The sodium acetate in water is the ionic solution in this project.
Phase Change- when a substance changes from one state to another. The three most common states are solid, liquid, and gas. One example of a phase change is ice (solid) melting into water (liquid).
Exothermic- when something is exothermic it releases or gives off energy. During the phase change, the hand warmer is releasing the energy it needed to separate the ionic compound making it exothermic.
Thermal Energy- A type of energy that is given as heat. All heat that exists is thermal energy. It is released when the phase change occurs in this project.
Energy Transfer- when energy changes from one type to another. All energy is conserved but can change between the different types. Some examples are mechanical, chemical, and electrical energy.
Baking Soda and Vinegar Bottle Rocket
Finally, we were given the opportunity to create our own project that reflected what we learned in this unit. I chose to design a baking soda and vinegar powered rocket, that would be able to reach up to 30ft. It was constructed out of a 16 oz. soda bottle, vinegar, baking soda packs(baking soda wrapped in paper towel to delay the onset of the reaction), and pencils/duct tape for aerodynamics and structural integrity.
Related Information
Lemon Battery Lab
|
In this lab, lemon mocks the chemical makeup of a battery when zinc and copper are added to it and can be used to generate power, but it generates a limited amount of said power. When a zinc and copper are added to a lemon, it closely mimics the makeup of a battery. All batteries are made of an anode, a cathode and an electrolyte solution. In the lemon, the zinc acts as the anode, or the negative end, the copper acts as the cathode or the positive side, and the lemon juice acts as the electrolyte or acidic solution. This means that by following the steps in the procedure, a lemon can obtain the three main components in a battery.
|
Cow Power
|
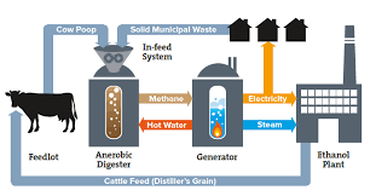
One of the first things that was discussed in this unit was about the power that can come from properly containing and chemical changing cow manure. In order to research this, we were given documents that discussed the negative affects on the environment that livestock creates and the ways that the livestock could be utilized in other ways to better the environment. The document also discussed the chemistry of how manure is turned into electricity. This process is shown in the diagram to the right.
|
Energy Transformations
|
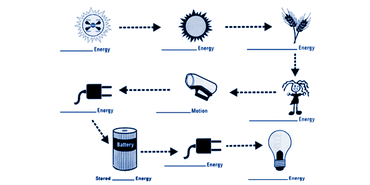
One of the main ways to understand the first law of thermodynamics (energy can not be created or destroyed, only transferred) is by visualizing how energy is transferred rather than created. We did this by completing and observing a Phet simulation that allowed us to electronically toy around with ways to generate and release energy. An example of one of the experiments is shown to the left, where a water wheel is used to light a light bulb. My partner and I used this specific simulation as inspiration for our project since we were taking a hypothetical route.
|
Reflection
I felt that the project was very strong overall, but all projects have their faults. One thing that I felt I did very well was staying motivated throughout the entire project. This project I really decided to be more determined and to follow through with my ideas and opinions, even if a global pandemic made things infinitely more difficult, especially considering that this entire project was conducted at home and through online learning. Another thing I feel I that I improved on from last time was my work ethic. In the last project, it was very difficult for me to stay on task, and caused a lot of problems in our group. Going into this project though my goal was to stay on task, but not be a killjoy at the same time. I feel I hit this goal right on the nose and did a really good job balancing productivity and and taking breaks throughout our time spent on this project. Although I did improve immensely over this project, there were several instances of unhealthy behaviors that I should've improved on.
One thing that I could improve on would be my stress management, for the most part I was relaxed throughout a project, but when you are locked in your house with your entire family and you're expected to uphold your same level of motivation, normalcy, and productivity that can send me over the edge sometimes. The second thing I would like to improve on, that I did poorly on during this project, would be my time management skills. It is already difficult to stay on task during a project with your team or teacher constantly urging you to continue to keep up, however with none of those people to help me there were several times where I fell behind on the project simply due to my lack of drive in completing the project earlier rather than later.
One thing that I could improve on would be my stress management, for the most part I was relaxed throughout a project, but when you are locked in your house with your entire family and you're expected to uphold your same level of motivation, normalcy, and productivity that can send me over the edge sometimes. The second thing I would like to improve on, that I did poorly on during this project, would be my time management skills. It is already difficult to stay on task during a project with your team or teacher constantly urging you to continue to keep up, however with none of those people to help me there were several times where I fell behind on the project simply due to my lack of drive in completing the project earlier rather than later.