Team: Heston Wilson
Introduction
In this project we were allowed the chance of making a work of art by utilizing the chemical processes of science and then turning it into a high quality art piece. The piece would later be placed in the new STEM building at San Marin, so it was absolutely essential to make sure that our fine art would be not only high-caliber, dazzling and enlightening; but also display an underlying message of our current STEM class. We were given a great deal of opportunity on the type of our work of art, so conceptualizing a plan was crucial to even begin this project.
Heston and I would later decide that making a piece of art that displayed the values of not only STEM Marin but also our message to future STEM classes. We decided to highlight this in our design, making it the centerpiece of which the rest of the design would revolve around. We accomplished this with two different chemical reactions: a double replacement reaction and a synthesis reaction. This would create not only a color change in the metal with the double replacement reaction, but the synthesis reaction would later be used as an additional reaction to outline our border and piece.
Heston and I would later decide that making a piece of art that displayed the values of not only STEM Marin but also our message to future STEM classes. We decided to highlight this in our design, making it the centerpiece of which the rest of the design would revolve around. We accomplished this with two different chemical reactions: a double replacement reaction and a synthesis reaction. This would create not only a color change in the metal with the double replacement reaction, but the synthesis reaction would later be used as an additional reaction to outline our border and piece.
Vocabulary
Synthesis Reaction: Multiple reactants combine to form a single product. A+B⟶AB
Decomposition Reaction: one reactant yields two or more products. AB⟶ A+B
Single Replacement: When an element replaces its like element in an ionic compound or acid. AB+C⟶AC+B. Cation+metal⟶cation+metal.
Double Replacement: The cations/anions change places between reactants and products. Forms a precipitate. AB+CD⟶CA+BD.
Combustion Reaction: Anytime anything burns, almost always exothermic (give off heat). Also, comes in contact with oxygen.
Precipitate: A solid that forms in a double displacement reaction, something that cannot be dissolved in an aqueous solution.
Insoluble: A substance cannot be dissolved in a liquid and cannot become part of a solution.
Phases of Matter: s,l,g= solid, liquid or gas. aq=dissolved in water. In a chemical equation, the phases of matter are listed as subscripts under the reactants and products.
Decomposition Reaction: one reactant yields two or more products. AB⟶ A+B
Single Replacement: When an element replaces its like element in an ionic compound or acid. AB+C⟶AC+B. Cation+metal⟶cation+metal.
Double Replacement: The cations/anions change places between reactants and products. Forms a precipitate. AB+CD⟶CA+BD.
Combustion Reaction: Anytime anything burns, almost always exothermic (give off heat). Also, comes in contact with oxygen.
Precipitate: A solid that forms in a double displacement reaction, something that cannot be dissolved in an aqueous solution.
Insoluble: A substance cannot be dissolved in a liquid and cannot become part of a solution.
Phases of Matter: s,l,g= solid, liquid or gas. aq=dissolved in water. In a chemical equation, the phases of matter are listed as subscripts under the reactants and products.
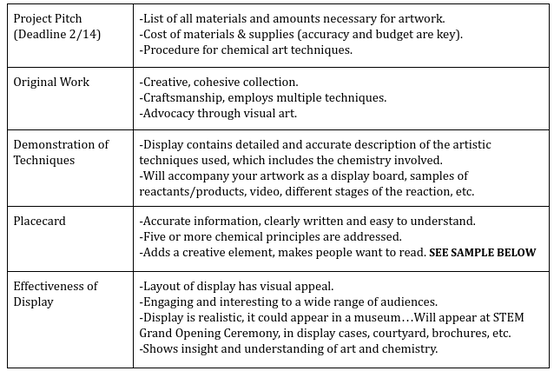
Project Guidelines
The driving question for this project was: "How can we use chemistry to design a work of art for our new STEM building?" and we were given this prompt:
"All artists are chemists. Artists understand and study the properties of specific materials and find ways to explore these properties to express views of themselves and the world around them. Art is the result of the human need to express ourselves. It tells stories of societies, eras, and individuals. Your challenge is to create a work of art that will be part of a four-piece cohesive collection to be displayed in our new STEM Science building. Throughout this unit we will focus on the different types of chemical reactions but it will be your task to incorporate this into a creative and timeless piece of art. The overarching theme is that chemistry is about change. Intertwined through each activity will be discussions of these changes of chemical interactions. It will be important to focus on the observable properties of the materials before and after the reaction and the atomic level explanation of what is occurring during a chemical reaction."
Listed above are the requirements we were given for this project. It was obvious that this was going to be a fairly open ended project, especially because the wide range of options available to us. This was hardly an obstacle though and we instead used this as a driving factor in focusing on our brainstorming for this project.
Listed above are the requirements we were given for this project. It was obvious that this was going to be a fairly open ended project, especially because the wide range of options available to us. This was hardly an obstacle though and we instead used this as a driving factor in focusing on our brainstorming for this project.
Chemical Processes
Our original idea was vastly different from what we actually ended up with at the deadline of our project. We originally planned to demonstrate the double replacement reaction of A.) The zinc in galvanized iron reacting to hydrocloric acid B.) The iron sheet reacting with Copper(II) Nitrate. The setback for this occured when we put too much Copper(II) Nitrate on the iron sheet which leaked through the masked tape reacting with the covered parts of our ornament.
Chemical equation: 1.) Zn(s) + 2HCl(aq) ----> ZnCl2(aq) + H2(g) 2.) Fe(s) + Cu(NO3) 2(aq) -----> Fe(NO3) 2(aq) + Cu(s)
In theory, this should have worked, but it went wrong when it came to the second reaction with the Copper(II) Nitrate. It was after this trial that we decided to add a secondary reaction by heating the iron sheet.
Since the Copper(II) Nitrate art didn't go as we thought, we went to researching reactions that would have substantial results on an iron sheet. Eventually we stumbled upon the same process by which metal smiths used to temper iron into steel, which not only makes the material we are working with stronger but it also changes the surface color of the iron. This would leave a more whitish ash color on the sheet instead of the standard dull gray.
The reaction that takes place when a flame is applied to a steel sheet follows:
2Fe(s)+ 3O2 (g)→2(Fe2)2O3
Chemical equation: 1.) Zn(s) + 2HCl(aq) ----> ZnCl2(aq) + H2(g) 2.) Fe(s) + Cu(NO3) 2(aq) -----> Fe(NO3) 2(aq) + Cu(s)
In theory, this should have worked, but it went wrong when it came to the second reaction with the Copper(II) Nitrate. It was after this trial that we decided to add a secondary reaction by heating the iron sheet.
Since the Copper(II) Nitrate art didn't go as we thought, we went to researching reactions that would have substantial results on an iron sheet. Eventually we stumbled upon the same process by which metal smiths used to temper iron into steel, which not only makes the material we are working with stronger but it also changes the surface color of the iron. This would leave a more whitish ash color on the sheet instead of the standard dull gray.
The reaction that takes place when a flame is applied to a steel sheet follows:
2Fe(s)+ 3O2 (g)→2(Fe2)2O3
Place card
LUKE VENEZIA - STEM Chemistry Possibility
Spring Collection 2020
To create this piece, galvanized iron and hydrochloric acid were first reacted to strip the zinc from the iron. This reaction produces hydrogen gas and zinc chloride. This leaves exposed iron. Finally, the iron reacts with cupric nitrate to form ferric nitrate in a single replacement reaction. Since iron is more reactive than copper the iron replaces the copper and forms a compound with the nitrate molecule. Add to this another reaction where heat from a bunsen burner is applied to the plate at a temperature of 590 degrees Fahrenheit. This will, therefore, change the physical color of the steel from its natural gray to a blue-purple.
These are the reactions described:
2Fe(s)+ 3O2 (g)→2(Fe2)2O3
Zn(s) + 2HCl(aq) ----> ZnCl2(aq) + H2(g) and Fe(s) + Cu(NO3) 2(aq) -----> Fe(NO3) 2(aq) + Cu(s)
Spring Collection 2020
To create this piece, galvanized iron and hydrochloric acid were first reacted to strip the zinc from the iron. This reaction produces hydrogen gas and zinc chloride. This leaves exposed iron. Finally, the iron reacts with cupric nitrate to form ferric nitrate in a single replacement reaction. Since iron is more reactive than copper the iron replaces the copper and forms a compound with the nitrate molecule. Add to this another reaction where heat from a bunsen burner is applied to the plate at a temperature of 590 degrees Fahrenheit. This will, therefore, change the physical color of the steel from its natural gray to a blue-purple.
These are the reactions described:
2Fe(s)+ 3O2 (g)→2(Fe2)2O3
Zn(s) + 2HCl(aq) ----> ZnCl2(aq) + H2(g) and Fe(s) + Cu(NO3) 2(aq) -----> Fe(NO3) 2(aq) + Cu(s)
One of the main requirements for this project was to create a place card. This card was to be displayed with the art piece that was created. Our group decided that we should create seperate place cards for both art pieces because it was a very cohesive project but also largely individual art pieces, and that place card is shown below.
The directions for the place card were pretty specific, but also gave creative freedom. The first requirement was to put our names so viewers could see who created each piece. The second was a title of the collection, which was "Spring Collection 2020". It was also supposed to show the chemical processes and look professional. The overall goal of these place cards were to let the viewers know that the art was created by chemists, and to do so, the place card had to catch people's eye. We used colors and bold lettering to tie the place card together and ensure that attention was drawn to it.
The directions for the place card were pretty specific, but also gave creative freedom. The first requirement was to put our names so viewers could see who created each piece. The second was a title of the collection, which was "Spring Collection 2020". It was also supposed to show the chemical processes and look professional. The overall goal of these place cards were to let the viewers know that the art was created by chemists, and to do so, the place card had to catch people's eye. We used colors and bold lettering to tie the place card together and ensure that attention was drawn to it.
Additional Information
Balancing Chemical Equations:
- Identify all of the elements in the equation.
- The number of each atom must be the same on either side of the equation.
- To make them balanced, a coefficient is added before the elements periodic symbol
- Balance the elements one at a time make sure that you change the coefficients and not the subscripts
- The subscripts show the charges for each element, and those also need to be balanced
- Balancing can also be done by creating a table with each element's amount and charge on each side of the equation
- Check your work to make sure that everything is balanced.
|
Solubility Guidelines:
The chart to the right was used all throughout this unit, mostly during work with double displacement reactions and other various chemical reactions. The solubility guidelines show what compounds will be soluble, and with what exceptions may apply to each. This table is used to predict if a precipitate would be created in a reaction. Although we did not use this in our art project, since our chemical reactions did not require the formation of a precipitate. Many people created paint, and to do so they created colored precipitates and mixed the products with a paint base. By using the solubility guidelines it would allow them to confirm that a precipitate would form in the reaction they were making. |
|
Reactivity Series:
This series of metals is in order from most reactive to least reactive, with most reactive on top and least reactive on the bottom. This table (left) is used in single displacement reactions. It is used to determine if a reaction will take place, and which metal will replace which. In a single displacement reaction, metal A will replace another metal B in a solution if A is higher up in the series. |
Finished Product
Above is a photo of the final art piece that our group completed. These art pieces make a statement about STEM Marin and the currennt STEM classes at San Marin, and were made with two reactions a double replacement and a synthesis reaction. In the STEM building, these pieces would be framed together with a place card and displayed in the newly completed STEM building.
Reflection
During this project everything that could have went wrong did, which made it extremely difficult to stay optimistic throughout the project's length. One thing that I felt I did very well was making up for lost time and staying on schedule. This project was very difficult for me mostly due to the fact that the first two days of the project, I was out sick at home meaning I lost two days during that time add to the fact that my teammate seemed very confused throughout this time leading to very little being done while I was out. However once getting back I made sure to communicate with my teammate on what needed to happen in the a lotted amount of time. Another thing I feel I that I improved on from last time was my empathy. In the last project, it was very hard for me to understand other peoples' struggles in a project, and caused a lot of problems in our group. Going into this project though both my teammate and I were hardly in the best position to complete this project but I understood this and because of that caused a great deal of tension within our group.
One thing that I could improve on would be my cooperation mostly with other groups, it became apparent very early on that our project was severely behind schedule and because of this we largely focused on purely our project. However this did lead to me ignoring many other classmates who needed either simple advice on certain areas of the project or needed certain materials from our group. The second thing I would like to improve on, that I did poorly on during this project, would be my ability to accept critique from other people. At many instances throughout this project many well intentioned people tried to give me feedback on the project and what I could do better. I have never been really good at accepting critique from people. But i would hope for that to change in the distant future.
One thing that I could improve on would be my cooperation mostly with other groups, it became apparent very early on that our project was severely behind schedule and because of this we largely focused on purely our project. However this did lead to me ignoring many other classmates who needed either simple advice on certain areas of the project or needed certain materials from our group. The second thing I would like to improve on, that I did poorly on during this project, would be my ability to accept critique from other people. At many instances throughout this project many well intentioned people tried to give me feedback on the project and what I could do better. I have never been really good at accepting critique from people. But i would hope for that to change in the distant future.